Material Science Research Using Electron Microscopy
- RIT/
- NanoImaging Laboratory/
- Research/
- Material Science Research Using Electron Microscopy
TEM and SEM Characterization of Alkali Activated Slag
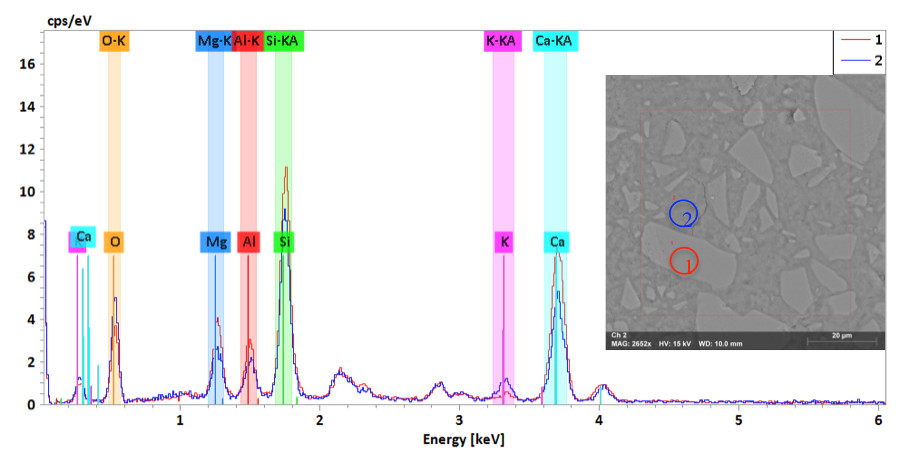
Figure 1. Slag activated with KOH SE images with EDS spectrum, curve 1 is the particle and curve 2 is the background.
Alkali-activated materials are considered third-generation binders after lime and ordinary Portland cement (OPC). There are two main families of alkali activated binders: the first one is the activation of industrial by-products such as fly ash and blast furnace slag with a mild alkaline solution and calcium silicate hydrate (C‑S‑H) as the main reaction product.
The second one is the alkali activation of natural aluminosilicates such as metakaolin with medium to high alkaline solutions. Alkali-activated materials have been known as an alternative to Portland cements due to the higher compressive strength, durability and environmental benefits [1,2]. Alkali‑activated blast furnace slag cements have been studied since the 1930s due to its high compressive strength, they can exceed 100 MPa in 28 days. The low Ca/Si ratio in slag improves its resistance to aggressive chemical materials such as acids, chlorides and sulphates [3,4,5]. Blast furnace slag is a highly heterogeneous material. It is well known that its chemical composition affects the physical properties of the alkali activated material, however there’s little work on how these inhomogeneities affect the microstructure and pore formation.
This study displays the characterization of two specimens; slag activated with KOH, and G5, which is slag with amorphous hydrophobic silica. Specimens were sputter coated with gold-palladium under a vacuum of 10 Pa, and then taken to TESCAN FESEM for examination. Backscattered electron (BSE) images were acquired at approximately 2.5kX using a 15kV beam voltage. In addition, elemental mapping and analysis were obtained by using energy dispersive X-Ray spectroscopy (EDS) on the specimens.
The SE image of the slag activated with KOH shows homogenous bright particles in a darker background. The elemental mapping indicates a high concentration of Ca, Si and Mg in the white particles and a low concentration in O, Al and K. On the other hand the background had a low concentration of Ca, Si and Mg, while O, Al and K are higher in the background, as shown in Figure 1. The spectrum of the bright particle and the background is shown in Figure 2 where Ca, Si and Mg peaks are higher in the particle as compared with the background. However, the Ca/Si ratios for both the particle and the background are relatively similar: 1.66 and 1.69, respectively. In contrast, G5 image shows homogenous dark gray particles in a lighter background. The elemental mapping of dark particles is similar to the bright particles in the slag specimen. It shows a high density of Ca, Si and Mg and a lower density of O, Al and K. The Ca/Si ratios are high (1.45 and 1.57) in the particles, while they are 1 and 1.27 in the background.
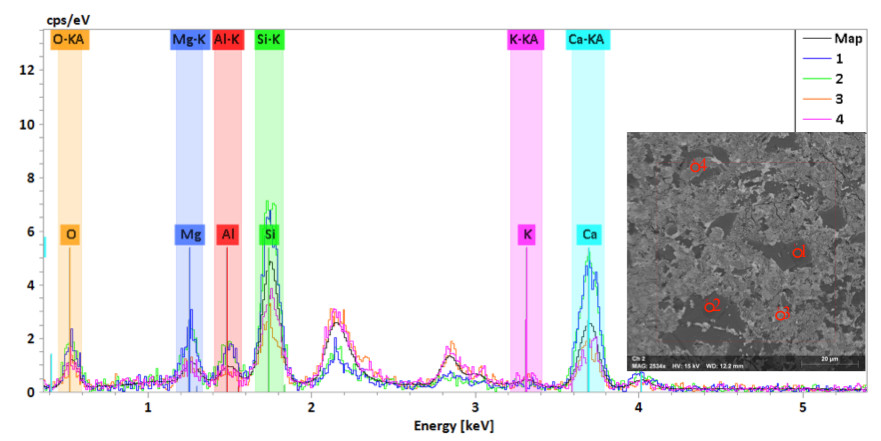
Figure 2. G5 SE images with EDS spectrum, curves 1, 2 are the particles, and 3, 4 are the background.
References
- [1] Li, C., Sun, H., & Li, L. A review: The comparison between alkali-activated slag (Si+ Ca) and metakaolin (Si+ Al) cements. Cement and Concrete Research, 40(9), 1341-1349 (2010).
- Pacheco-Torgal, F., Castro-Gomes, J., & Jalali, S. Alkali-activated binders: A review: Part 1. Historical background, terminology, reaction mechanisms and hydration products. Construction and Building Materials, 22(7), 1305-1314 (2008).
- Xu, H., Provis, J. L., van Deventer, J. S., & Krivenko, P. V. Characterization of aged slag concretes. ACI Materials Journal, 105 (2) (2008).
- Shi, C., Jiménez, A. F., & Palomo, A. New cements for the 21st century: the pursuit of an alternative to Portland cement. Cement and Concrete Research, 41 (7), 750-763 (2011).
- San Nicolas, R., Bernal, S. A., de Gutiérrez, R. M., van Deventer, J. S., & Provis, J. L. Distinctive microstructural features of aged sodium silicate-activated slag concretes. Cement and Concrete Research, 65, 41-51 (2014).