Research

Drug Delivery and Pharmacokinetics
Advanced biomedical disease treatment therapies that restore function and reduce symptoms require carefully timed and dosed, site-directed delivery of drugs, gene vectors and/or chemical compounds over a period of time. Pioneering studies in gene therapy and drug treatments have produced exciting results towards protection and regeneration of organ systems, including neural and sensory systems. Of particular interest is hearing loss, where first-year treatment costs in the US are projected to rise to over $54 billion by 2030. For the profoundly deaf, advances in cochlear implant (CI) technology with co-delivery of agents that preserve neural function are critically needed, especially for the growing pediatric population. Our research is addressing these challenges through novel integrated micropumps for murine drug delivery, quantitative pharmacokinetic models to predict spatio-temporal cochlear drug concentrations, and integration of these technologies with CI technology to accelerate translation to human clinical use.

Implantable Micropumps
Advances in protective and restorative biotherapies have created new opportunities to use site-directed, programmable drug delivery systems to treat auditory and vestibular disorders. Our lab has created an implantable micropump platform with wireless control via additive manufacturing of micropump actuators directly around a microcatheter tubing on the back of a printed circuit board assembly. 3D printing enabled ultra-thin microreservoirs to be integrated with the micropump providing a complete, biocompatible system for peristaltic delivery of drugs with nanoliter per minute flow control in a form factor suitable for subcutaneous implantation in mice, and future translation to human clinical use.
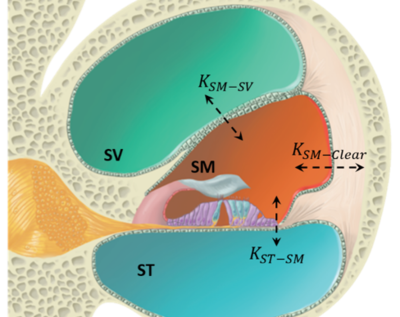
Pharmacokinetic Models
Pharmacokinetic models of inner ear drug delivery are advanced by combining non-invasive micro-CT imaging with a simulation-prediction paradigm to learn transport parameters governing distribution and clearance of drug within the cochlea. These models leverage 3D image registration to a labeled cochlear atlas, enabling extraction of the spatio-temporal concentration profiles during delivery of a contrast agent to the cochlea of small rodents. The resulting pharmacokinetic model predicts concentrations within all major compartments of the cochlea with user control of the drug delivery profile.

Cardiac Physiology, Modeling and Monitoring
Cardiovascular disease impacts nearly 30% of those over age 65 and is the leading cause of death in the United States. Each year there are over 710,000 heart attacks, with nearly 74% a first heart attack. The high direct medical care and indirect costs of cardiovascular disease approached $450 billion a year in 2010 and are projected to rise to over $1 trillion a year by 2030. The hospitalization rate for heart failure (HF) patients has more than tripled in persons age 45 and older in the past 3 decades, with 1 million hospitalizations each year at a cost of $12 billion. The NIH estimates nearly 50% of heart failure admissions may be avoidable with proper management, requiring a shift from ineffective episodic office-based management by physicians. Our research aims to transform the way cardiovascular disease is managed to improve outcomes, quality of life, and dramatically reduce cost. First principle models of hemodynamics are providing new insights that enable advanced, in-home and wearable cardiovascular monitoring systems for comprehensive cardiovascular assessment. Signal processing and automated intelligence are at the heart of these systems yielding clinical-grade measurements with minimal patient interaction.
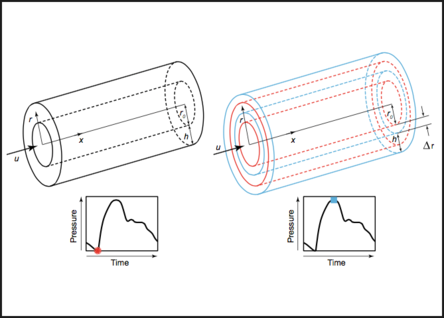
Cardiovascular Modeling
This research aims to understand pulse wave propagation in the arterial system using first principles modeling of hemodynamics. The pulse wave velocity is commonly used as a clinical marker of vascular elasticity, however multiple physiologic and arterial parameters impact this measure potentially confounding interpretation of results. Our work integrates convective fluid phenomena, hyperelastic constitutive relation, finite vessel deformation, radial motion, and inertia of the wall in addition to peak pressure, ejection time, and ejection volume. The work leads to a solution of the inverse problem of hemodynamics and lays the foundation for continuous, non-invasive blood pressure monitoring based on pulse wave velocity.

Biomedical Signal Processing
Our lab develops advanced biomedical signal processing techniques for power savings, robust extraction in noise, and trend determination including Kalman filtering, PCA, compressed sensing and machine learning. Of specific interest are techniques for the electrocardiogram (ECG), photoplethysmogram (PPG) and ballistocardiogram (BCG). Application domains are explored from fetal ECG extraction, to robust QT interval determination for drug studies, to true sub-Nyquist sampling for radical power savings in the PPG, and to precise feature delineation in noise plagued signals such as the BCG.
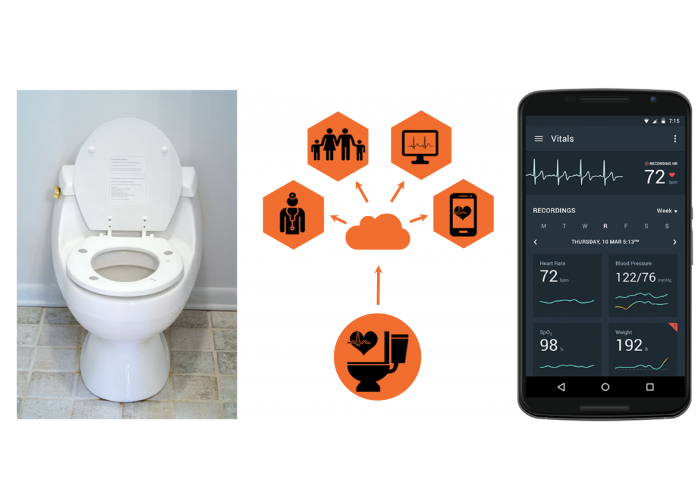
Non-Invasive Physiologic Measurements
This research aims to address patient adherence challenges of in-home monitoring through innovative integration of physiologic monitors into non-traditional platforms. Our objective is to detect changing health and deterioration to enable early interventions to improve quality of life and reduce hospitalizations. A toilet seat–based cardiovascular monitoring system integrating the electrocardiogram (ECG), ballistocardiogram (BCG), and photoplethysmogram (PPG) enables extraction of key cardiac parameters including heart rate, blood pressure, weight, cardiac output, blood oxygenation, HR variability, QRS duration, QTc interval, arrhythmias, and aortic stiffness. This system is being evaluated in clinical studies to reduce hospitalization rates for heart failure subjects, and to detect early cardio-toxicity from thoracic radiation therapy.

Sensors and Devices
Sensors, transducers, and full microsystems provide solutions to a broad range of challenges from biomedical research to healthcare, our military and the internet of things. Microelectromechanical Systems (MEMS) technologies have traditionally been at the heart of these devices, but emerging technologies including additive manufacturing create opportunities to develop sensors and transducers on a broader range of substrates and with novel device characteristics. The continued miniaturization of commercial off the shelf electronics with commensurate power savings and enhanced computational power enables new classes of wearable sensors and remote monitoring capabilities that integrate advanced algorithms and are wirelessly interrogated. Our research is creating new classes of materials, fabrication process capabilities and devices for biomedical applications spanning molecular and cellular interactions to environmental monitoring.
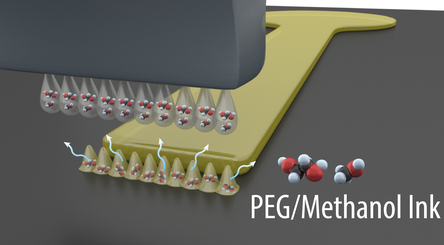
Direct Write Sensors, Actuators, and Electronics
Additive manufacturing approaches are leveraged for advanced thin film sensors, actuators and electronics on flexible substrates and over classic MEMS devices. Nano-inks are printed using aerosol jet, ink-jet, and microdispensing systems in nano to micro layer film thicknesses on non-planar substrates. Photonic sintering technologies enable rapid sintering of materials on low temperature substrates, critical for integration over MEMS that incorporate polymer materials. Specific areas of investigation include piezoelectric sensors and actuators, and commercial off the shelf electronics integration on flexible polymer substrates and MEMS micropumps.

Sensors to Combat Traumatic Brain Injury
The Blast Gauge® System is a wearable technology designed to measure overpressure exposures in blast events for the military. This DARPA funded effort led to deployment on over 11,000 troops in Afghanistan providing a new capability in measuring individual exposures for our service members. The system revealed significant occupational exposures from heavy weapons firing; insight that has led to legislation and DoD monitoring programs. The system has been commercialized by BlackBox Biometrics, Inc. (https://blastgauge.com/).
Sponsors






