Connelly Lab
Contact
Sandi Connelly
Principal Investigator
sjcsbi@rit.edu
Overview
The Connelly lab is an aquatic ecology lab with a focus on anthropogenic and natural stressors in freshwater systems. We are a relatively small lab group with extensive collaborations within RIT in chemistry, sustainability, and engineering, and outside of RIT.
Learn more about Dr. Sandra Connelly.
Projects
This area of research has been a passion of mine since I first worked in freshwater systems in extreme environments during my graduate career. I have found that it is also an area of work that resonates with students, can be applied to a multitude of aquatic systems, and is relative inexpensive to support while giving high rewards to the participants (vast research experiences and the opportunity for dissemination of work through presentations and manuscripts). Currently I have eight undergraduate students of various biology and chemistry declared majors in my lab that are addressing some aspect of UV-induced effects on organisms. Many have gone far beyond the simple “survival” of organisms and are more interested in the overall fitness of the populations (survival and reproduction in stressed systems) and the anthropogenic effects on the natural systems that may alter the responses of the organisms. This is work that I see continuing for the duration of my career in some way, including my most recent established collaboration with Dr. Tim McDermott at Montana State University, in which we will investigate the rate of DNA damage and repair in extremophiles living in hot springs in Yellowstone National Park (grant submission to NASA Exobiology, July 2016).
I have only recently become involved in research investigating the ecological impacts of crumb rubber substrates from artificial athletic fields. Very little work has been done on the leaching of substances from the crumb rubber (recycled tires) that is not directly related to human health. By this, my research group is focusing on the terrestrial and aquatic impacts of these artificial turf fields. Currently, I have two undergraduate students working on this project (1 chemist, 1 biologist), investigating the leachate and runoff in self-contained mesocosms. The collaborative nature of this type of work, and multiple levels of study from biology and chemistry, permit a very dynamic research group (faculty, undergraduate, and graduate students). We are in the infant stages of this project currently, developing new techniques, modifying published methods to better fit our environmental test conditions, and using some well established techniques to study population effects (fitness = survival and reproduction in the given conditions). Dr. Jeff Mills (Chemistry, RIT) is testing and designing new protocols to identify the leachate components, and working to isolate those components for individual and composite ecotoxicology testing. Dr. Andre Hudson (Life Sciences, RIT) is assisting with identifying changes in the terrestrial bacterial communities and how such changes will impact trophic levels in those systems, directly and indirectly. This type of collaboration and breadth of the project itself could allow for extensive collaborations with the involved labs, and lead to very high successes of scholarship dissemination and funding in the future. Further, I am working with Dr. Callie Babbitt (Sustainability, RIT) to develop tools and techniques for assessing the toxicity of recycled battery components in ecosystems. Currently, I am mentoring her PhD student (Elizabeth Wronko) on this project, and we continue to expand the investigation in to the effects of C70 and C60 nanomaterials on algae, freshwater invertebrates, and trophic level interactions. The National Science Foundation currently funds this work and we are seeking additional funding from the US Environmental Protection Agency to expand this work in the future.
We cannot forget that the application of the knowledge and the dissemination of research knowledge to our students can be some of the most powerful teaching tools. The general biology course redesign that I undertook in Spring 2014 (see Scholarship Statement) led me to consider other possible avenues to improve the general education science curriculum, and improve the accessibility of the curriculum to under-represented groups (targeting deaf and hard-of-hearing students). In November 2015, I submitted a National Science Foundation Division of Undergraduate Education grant with Dr. Matt Lynn (RIT) in which we propose to develop tools to aid deaf and hard-of-hearing students in their transition from Associate Degree programs to STEM Baccalaureate programs. While this grant was not funded in the last round, it was very favorably reviewed and it will be resubmitted in November 2016. Further, I continue to engage in collaborations with colleagues within and external to RIT in the area of pedagogy and instructional design. I have developed materials with instructors that we now use jointly in our classes at our own institutions. These types of collaborations are critical for the future of education in an ever changing technological and preparedness arena that is the classroom! These collaborations will lead to presentations, publications, and external-funding opportunities in the foreseeable future.
Lab Resource
Daphnia Genomics Consortium
Students
|
Andrew Layman Andrew has been working on the crumb rubber project since 2015 and has presented at three conferences/symposia, including the Rochester Academy of Science and the Association for the Sciences of Limnology and Oceanography Annual Meeting (Santa Fe, NM). Andrew's work focuses on the isolation and quantification of leachate constituents from the crumb rubber. |
 |
|
Alexandra Mace Alex was a RIT COS Summer Undergraduate Research Fellow in the summer 2016. Her work focused on the first trials of crumb rubber in terrestrial systems, determining the toxicity of the crumb rubber on earthworms and isopods. She presented at the RIT Undergraduate Research Symposium in August 2016. |
 |
Where are they now?
Lindsay Smith
Research proposal pursued: UV radiation causes extensive damage and mutation to an organism’s DNA, and with an increases of UV levels in freshwater ecosystems, it is important to explore the effects this phenomenon is causing. This project will focus on the extent of damage and ability of DNA repair after Daphnia species experience exposure to UVA and UVB radiation. The Various daphnia species have been exposed to different types and duration of UV radiation. DNA samples were taken before exposure, immediately after exposure, and after a recovery period. Now that exposures and DNA has been collected, the work of this semester will be to develop an ELISA protocol to measure the amount of mutations in each sample and compare these values. The knowledge obtained from Daphnia can be used as a model for freshwater ecosystems as a whole. Knowledge of UV radiation’s effects will be significant in understanding changes in freshwater ecosystems that may occur as environmental changes continue. It is important to understand not only survival and mutation rates of organisms in the presence of UV but to also the ability to recover. This data will illustrate the varying abilities to repair and levels of adaptability of different species of daphnia as they face increasingly high amounts of UV radiation in their freshwater ecosystems.
Kate Meier
Research proposal pursued: Objectives: Determine the anti-predator defenses of Jefferson salamanders on RIT campus and observe correlations between salamander length, color, flight response, tail lift. Kate studied the anti-predator behavior of RIT Jefferson salamanders to find unique relation between their tail responses and physical attributes. She located salamander habitats on RIT campus and performed field data collection of: salamander location, quantity of salamanders at specific site, initial uncovering response (stationary or flee), perform predator behavior stimuli (three count tap), measure body proportions, photograph salamanders for coloration scale. She quantified salamander tail responses include: tail lift below 45 degrees, tail lift above 45 degrees, wriggle, a combination of lift and wriggle, or none and performed statistical analysis of data to determine exact relationship of anti-predator responses. Kate is a co-author on a manuscript in review at American Biology Teacher (June 2016).
Zach Kopp & Robby Keith
Research proposal pursued: Zach and Robby worked together on the following project: Problem: Understanding the role of Vitamin D in aquatic ecosystems, particularly the connection between algae and microinvertebrates.
The study of vitamin D3 sequestration in Pseudokirchneriella algal biomass will assist collaborative research efforts involving the bio-indicator species, Daphnia pulex. Previous work at the RIT research labs of Connelly, Tan, and Cody has been focused on exposing Daphnia to UV radiation and monitoring the photo-properties of vitamin D3 or the internalized metabolite, 25-hydroxyvitamin-D3. This work will be a continuation of the prior experimental trials, involving the use of fluorescent-labeled compounds of Vitamin D (Cody lab). Previous experimentation showed that under the stress of UV-A, the algae was shown to be hungry for a much larger proportion of the added vitamin D3 than that of the Daphnia. Prior studies have shown that there is a more complex role than expected with the mitigation involving the system of antioxidant uptake and UV irradiation by algae in an aquatic system. Experimentation will include multiple varieties of algal species. Photos of selected algae species are to be taken, in vivo, to determine the location of the vitamin sequestration in the algal body. This continuation is a critical expansion of previous phases of this project and will aid in determining the effects of vitamin D3 on the aquatic ecosystem. Zach and Robby are co-authors on a manuscript published in PLOS ONE (http://journals.plos.org/plosone/article?id=10.1371%2Fjournal.pone.0131…;
Steven Wilbert
Steven imaged Daphnia as part of the vitamin D3 project using scanning electron microscopy (SEM). He is a co-author on a manuscript published in PLOS ONE (http://journals.plos.org/plosone/article?id=10.1371%2Fjournal.pone.0131…;
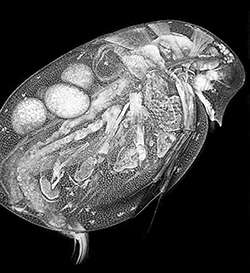
Kelly Walling
Research proposal pursued: Increased levels of ultra-violet radiation have been shown to have detrimental effects on numerous organisms. Therefore, the possibility of photo-protective compounds is increasingly appealing. The focus of this project is the potential photo-protective properties of vitamin D3 in the freshwater micro-crustacean, Daphnia. I hypothesize that vitamin D3 will increase the fitness (survival and reproduction) of Daphnia exposed to UV radiation, both long wave or UV-A, which emits at 400-315nm, and medium wave or UV-B, which emits at 315-280nm. Kelly was part of a collaborative research project that included Dr. Connelly, Dr. Cody, and Dr. Tan labs at RIT. Her side of the project revolves around studying the Daphnia. During summer of 2011 she ran experiments testing the overall health and viability of Daphnia pulicaria under UV and non-UV conditions. Daphnia spp. were raised on fixed concentrations of vitamin D3 (0- 3,000 ng ml-1) and exposed to UV-A radiation (0 - 60 kJ m-2). Control populations (ones with no UV exposure) were compared to populations under variable conditions. The data conclusively showed that Daphnia given vitamin D3 could survive higher doses of UV-A radiation. Her focus was on the effects of vitamin D3 in respect to UV-B exposure. Her project was significant because reduction in Daphnia populations have been shown to have higher food web impacts. Kelly is a co-author on a manuscript published in PLOS ONE (http://journals.plos.org/plosone/article?id=10.1371%2Fjournal.pone.0131…;
Maria Tobias
Research proposal pursued: The continual depletion of the Earth’s stratospheric layer has led to a higher amount of ultraviolet (UV) rays reaching the Earth’s surface 1, severely affecting the global climate, and forcing many organisms to develop mechanisms to survive.2 UV radiation is absorbed by DNA in all organisms and can cause lethal genomic mutations. Because organisms cannot survive high levels of DNA damage, populations must find ways to protect themselves. An organism’s ability to survive stressors will depend on the types and number of mutations induced in their genome. In freshwater organisms, physiological and behavioral changes such as migrating (swimming) away from areas of higher UV exposure or increasing their DNA repair rates to compensate for increased damage, will be triggered by internal mechanisms.2
Daphnia, a freshwater microcrustacean, is a particularly good study organism because they are transparent (potential for high levels of direct DNA damage), are easy to rear in a lab, and are found in most freshwater systems in nearly all climates. Daphnia are parthenogenic organisms (females asexually reproduce female offspring)3, providing a unique opportunity to observe the behavioral and physiological changes in these organisms over many generations without high levels of genetic variability that would be typically observed in sexual organisms. Food availability is known to have significant effects on growth rates in Daphnia, so the appropriate amount of the algal food source was determined to maintain a high fitness level (survival and reproduction). The amount of the algae Selenastrum required to survive and reproduce steadily is 2.0mL D. magna reproduction appear to be unaffected by the different UV-A treatments. D. magna appear to be unaffected by the acute UV-A dose to which they were exposed; D. pulicaria appears to be affected by the dose; D. pulex is significantly affected by increasing acute UV-A in this experiment under both food types.




